The integrated solution for cancer diagnosis and treatment covers all aspects of the cancer care continuum,from screening and diagnosis to treatment and rehabilitation.With the development and application of big data,artificial intelligence,and new medical materials,the integrated solution for cancer diagnosis and treatment will leverage these technologies and materials to conduct in-depth mining and analysis of a large number of case data.It will provide a comprehensive,integrated,continuous,and industry-leading solution for the entire cycle of cancer diagnosis and treatment.This approach aims to improve the effectiveness of cancer diagnosis and treatment,reduce medical costs,enhance patient experience,and ultimately contribute to the extension of patients'survival periods.
Respiratory interventional system
-
MultiStage® Balloon Dilatation Catheter (Pulmonary) employs a three-in-one design, allowing for continuous and progressive dilation. Indicated for the dilatation of respiratory tract stenosis during endoscopic procedures. Please refer to the corresponding product registration/filing certificate in each sales region for specific product information.



-
The Zephyr endobronchial valve is a one-way duckbill valve with a support frame made of Nitinol and a silicone rubber coating. It is placed into the bronchus through a dedicated delivery catheter and is used to control airflow to improve lung function and reduce air leakage in patients with emphysema with uneven lesion distribution.For detailed product information, the official product registration/licensing documentation for the respective market should be consulted.



-
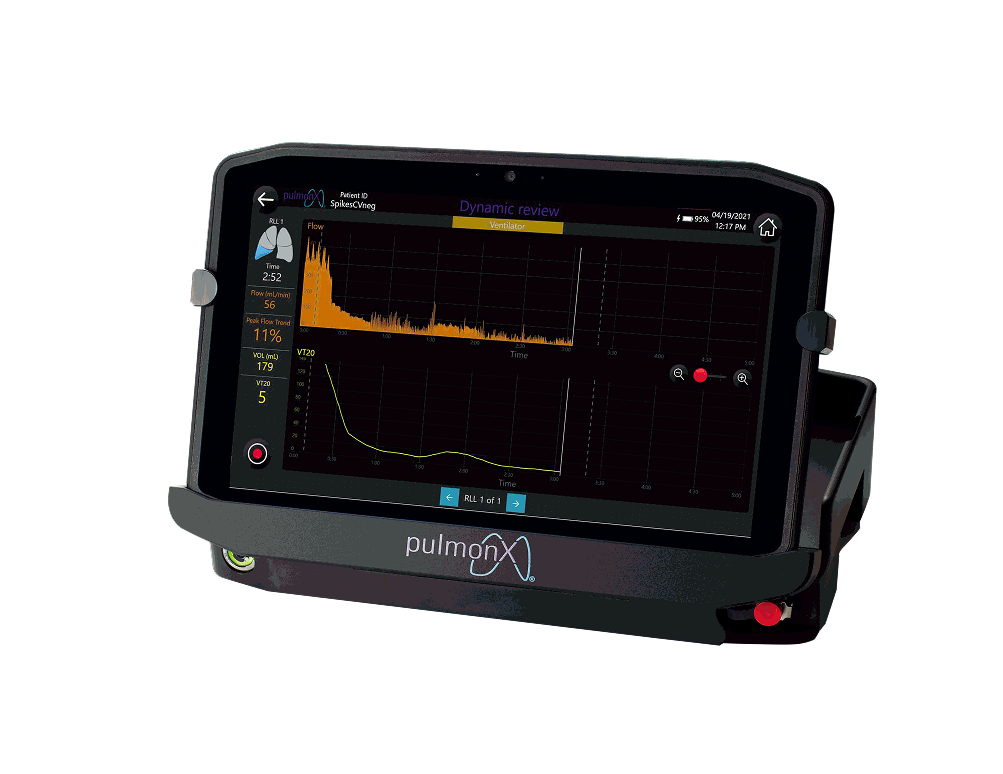
The Chartis Pulmonary ventilation function tester is suitable for the bronchoscopy of patients. It is used to check whether there is bypass ventilation between the pulmonary lobes of patients with COPD and emphysema through the supporting special catheter and fittings and other consumables. The Chartis® System provides precise flow and pressure readings for specific lobes in the lung to assess absence of collateral ventilation, which is a predictor of response to treatment.For detailed product information, the official product registration/licensing documentation for the respective market should be consulted.


-

 MultiStage® Balloon Dilatation Catheter (Pulmonary)
MultiStage® Balloon Dilatation Catheter (Pulmonary)
-

 Zephyr endobronchial valve
Zephyr endobronchial valve
-

 Chartis Pulmonary ventilation function tester
Chartis Pulmonary ventilation function tester
Gynecological system
-
Lifusion GC 100 lectric Gynecological Treatment Chair is a premium electrictreatment chair designed for gynecological examination and treatment which is more in line with the physiological structure of Asian women. It has electric adjustment and position memory function for easier operation.It pro-vides clinicians with clear diagnosis and treatment vision. Ergonomics design reduces patient tension.For detailed product information, the official product registration/licensing documentation for the respective market should be consulted.



-
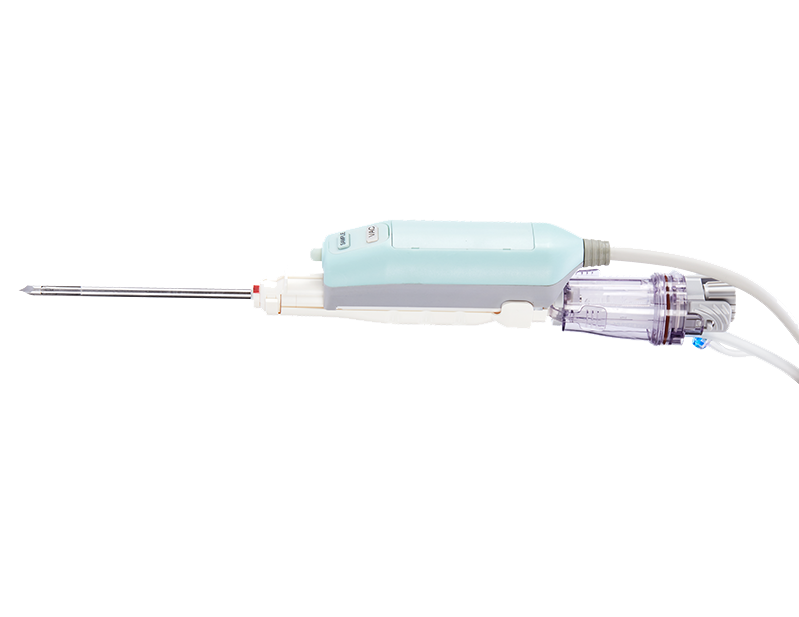
Lifusion disposable breast biopsy probe, used in conjunction with mainstream breast biopsy system such as BD EnCor EnspireTM Breast Biopsy System, is indicated to completely or partially remove the imaged breast abnormalities under the guidance of an ultrasound system for biopsy sampling. For detailed product information, the official product registration/licensing documentation for the respective market should be consulted.


-

 Lifusion GC 100 Electric Gynecological Treatment Chair
Lifusion GC 100 Electric Gynecological Treatment Chair
-

 Disposable Breast Biopsy Probe
Disposable Breast Biopsy Probe
Urology system
-
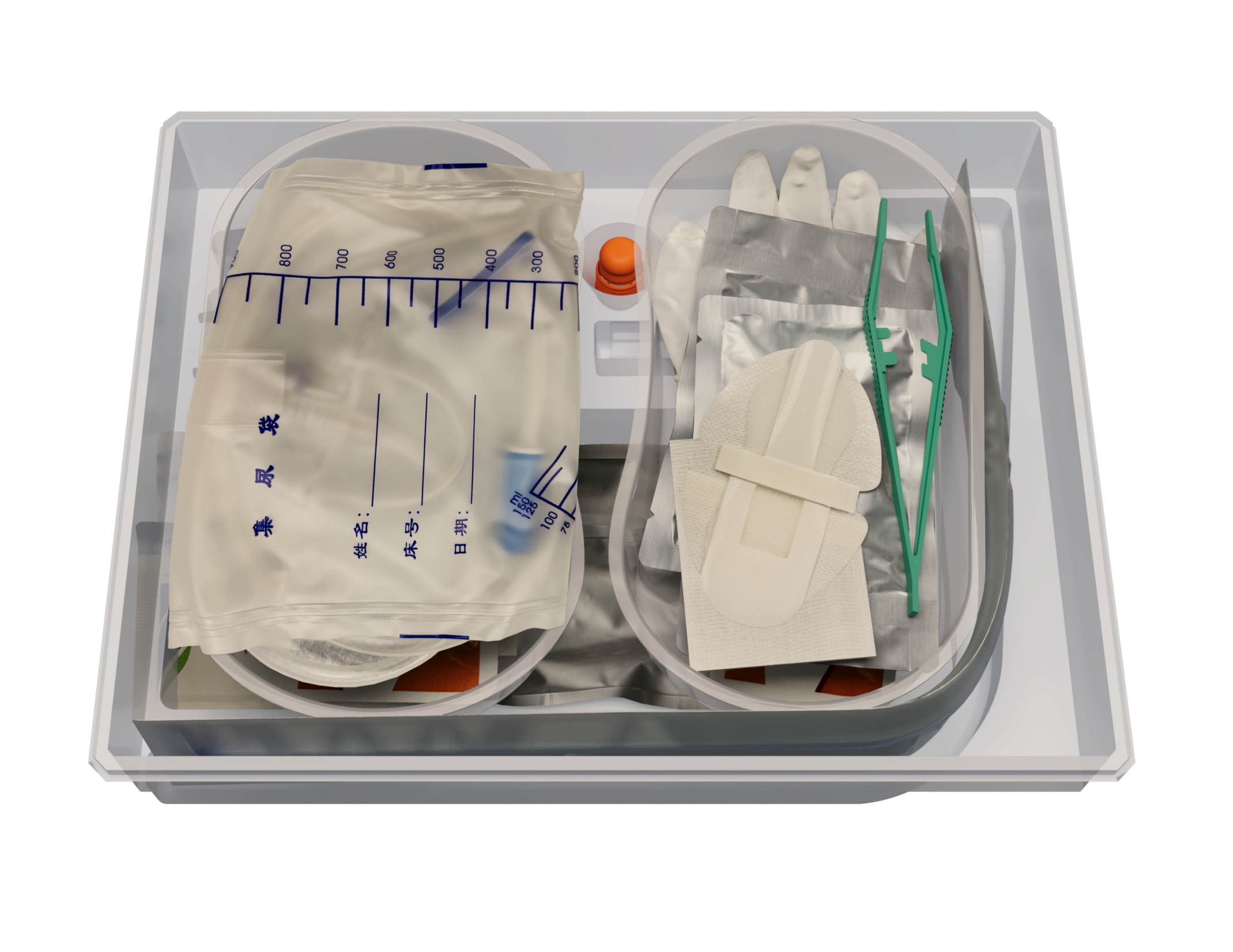
Single-use sterile ultra-slippery silicone catheterization kit belongs to the new generation of high-grade medical consumables for clinical catheterization. Superlubricated coating , persistent lubrication,long-term antibacterial effect. Medical-grade silicone, 28 days in Vivo.For detailed product information, the official product registration/licensing documentation for the respective market should be consulted.


-
Single-Use Sterile Silicone Urinary Catheter belongs to the new generation of high-grade medical consumables for clinical catheterization. This product has undergone dual sterilization through irradiation and EO, and can meet the needs of catheterization operations in various clinical scenarios.For detailed product information, the official product registration/licensing documentation for the respective market should be consulted.



-

 Single-use sterile ultra-slippery silicone catheterization kit
Single-use sterile ultra-slippery silicone catheterization kit
-

 Single-Use Sterile Silicone Urinary Catheter
Single-Use Sterile Silicone Urinary Catheter
Universal diagnosis
-
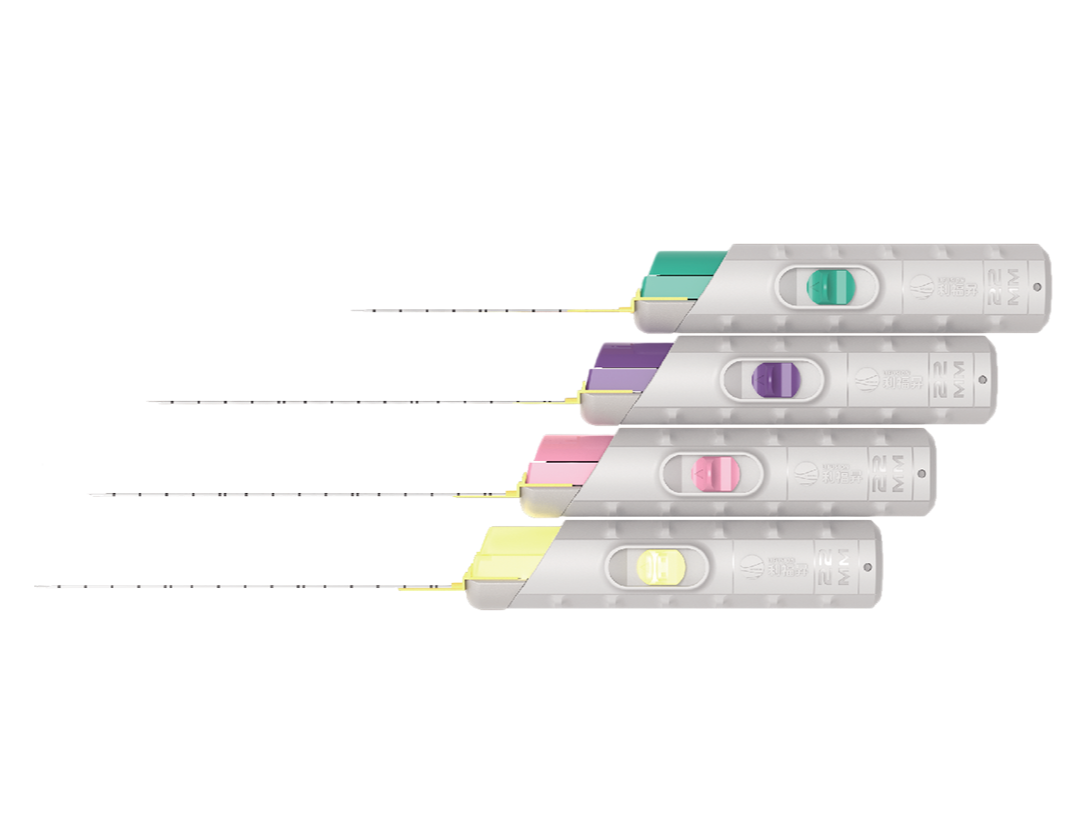
Lifusion Disposable Core Biopsy lnstruments are used to obtain specimens from human tissues such as liver, kidney,prostate, spleen, lymph nodes, and soft tissue tumors for biopsy.For detailed product information, the official product registration/licensing documentation for the respective market should be consulted.


-

 Lifusion Disposable Core Biopsy Instrument
Lifusion Disposable Core Biopsy Instrument
Tumor vascular access system
-
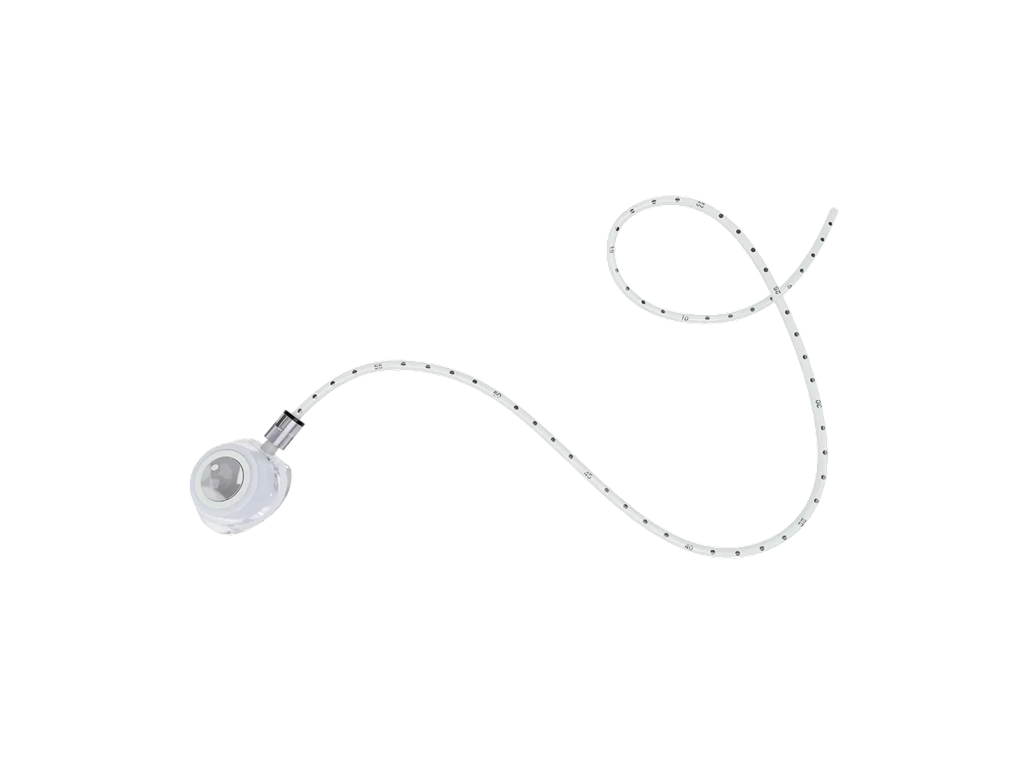
The Implantable Drug Delivery Device and Accessories (PORT) is intended for the treatment of patients who require repeated vascular access in clinical practice. It can also be used for injecting drugs, intravenous infusion, intravenous injection of drug solutions and blood products, as well as for collecting blood samples. Please refer to the product registration/filing certificate in each sales region for specific product information.



-
The Implantable Drug Delivery Device and Accessories (PORT) is intended for the treatment of patients who require repeated vascular access in clinical practice. It can also be used for injecting drugs, intravenous infusion, intravenous injection of drug solutions and blood products, as well as for collecting blood samples. Please refer to the product registration/filing certificate in each sales region for specific product information.


-
The Peripherally Inserted Central Catheter and Accessories (PICC) is intended for short-term (under 30 days) or long-term (over 30 days) peripherally inserted central venous access for intravenous therapy or blood collection. Please refer to the product registration/filing certificate in each sales region for specific product information.



-
The Implantable Drug Delivery Device (PORT) is intended for the treatment of patients who require repeated peripheral venous access in clinical practice, including injection of drugs, intravenous infusion, blood products (including blood cell components and plasma components), collection of blood samples, and high-pressure infusion of contrast agents. Please refer to the product registration/filing certificate in each sales region for specific product information.



-

 Implantable Drug Delivery Device and Accessories(PORT)
Implantable Drug Delivery Device and Accessories(PORT)
-

 Pulmonary Artery Balloon Dilatation Catheters
Pulmonary Artery Balloon Dilatation Catheters
-

 Peripherally Inserted Central Catheter and Accessories (PICC)
Peripherally Inserted Central Catheter and Accessories (PICC)
-

 Implantable Drug Delivery Device(PORT)
Implantable Drug Delivery Device(PORT)
Pulmonary vascular intervention system
-
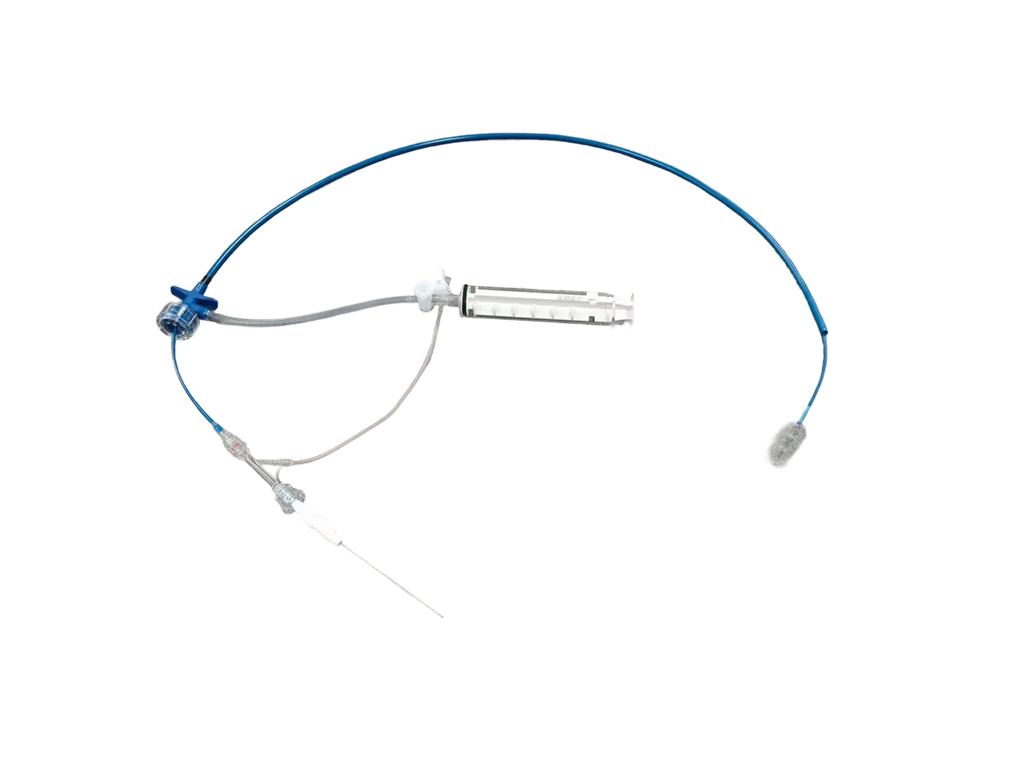
The Ruifan® Pulmonary Artery Thrombectomy Stent System is indicated for transcatheter thrombectomy in patients with acute high-risk pulmonary embolism or intermediate-risk pulmonary embolism with clinical deterioration under one of the following conditions: (1) Patients with thrombosis in the main trunk or major branches of the pulmonary artery who are at high risk of bleeding or have contraindications to thrombolysis; (2) Patients with thrombosis in the main trunk or major branches of the pulmonary artery who have not responded to thrombolysis or aggressive medical treatment. Please refer to the product registration/filing certificate in each sales region for specific product information.


-
The Pulmonary Artery Balloon Catheter is intended for adult patients with chronic thromboembolic pulmonary hypertension who are not candidates for pulmonary artery endarterectomy, as well as for adult patients with residual or recurrent pulmonary hypertension after pulmonary artery endarterectomy, for dilatation during percutaneous transluminal pulmonary angioplasty. Please refer to the product registration/filing certificate in each sales region for specific product information.



-
The Pulmonary Artery Guiding Catheter is indicated for establishing access for interventional therapy and for introducing interventional or diagnostic devices into the target site within the pulmonary artery. Please refer to the product registration/filing certificate in each sales region for specific product information.



-
The Pulmonary Artery Microcatheter is intended for selectively delivering liquids or agents to the target site within the pulmonary artery during diagnosis and/or treatment. Please refer to the product registration/filing certificate in each sales region for specific product information.



-

 Ruifan® Pulmonary Artery Thrombectomy Stent System
Ruifan® Pulmonary Artery Thrombectomy Stent System
-

 Pulmonary Artery Balloon Dilatation Catheters
Pulmonary Artery Balloon Dilatation Catheters
-

 Pulmonary Artery Guiding Catheter
Pulmonary Artery Guiding Catheter
-

 Pulmonary Artery Microcatheter
Pulmonary Artery Microcatheter
















